Sometimes a paper begins with a question. Other times, it begins with discomfort.
In this case, the discomfort came from feeling that we might be looking at the system too simply. We did not want to stop at a checklist of helminths or at a basic comparison between rainy and dry seasons. We wanted to understand something more ecological: how an entire parasite community changes when water, landscape, and therefore transmission opportunities change.
That was what led us into this study on the gastrointestinal helminth fauna of Kinosternon scorpioides on Marajó Island. It was also one of the most meaningful parts of my contribution to the paper: helping shape the analysis and discussion so that the ecological story behind the data became clearer.
We were not only looking for “more” or “fewer” parasites
From the start, we knew that Amazonian seasonality is not just background scenery. In systems such as Marajó, the alternation between rainfall and flooding on one side and low-water conditions on the other reshapes ecological interactions. It changes habitat availability, connectivity, and the chances of encounters among hosts, parasites, and intermediate hosts. That idea sits at the core of the paper.
What we found was revealing. Overall prevalence remained high, but total parasite load dropped by 71% during the dry season. More importantly, community composition shifted: some species were associated with the rainy season, while others increased during the dry season. The result was not simply a linear reduction in parasitism, but a restructuring of the parasite community.
And that is where a central idea of the paper emerged: temporal niche partitioning.
The result that made us think the most
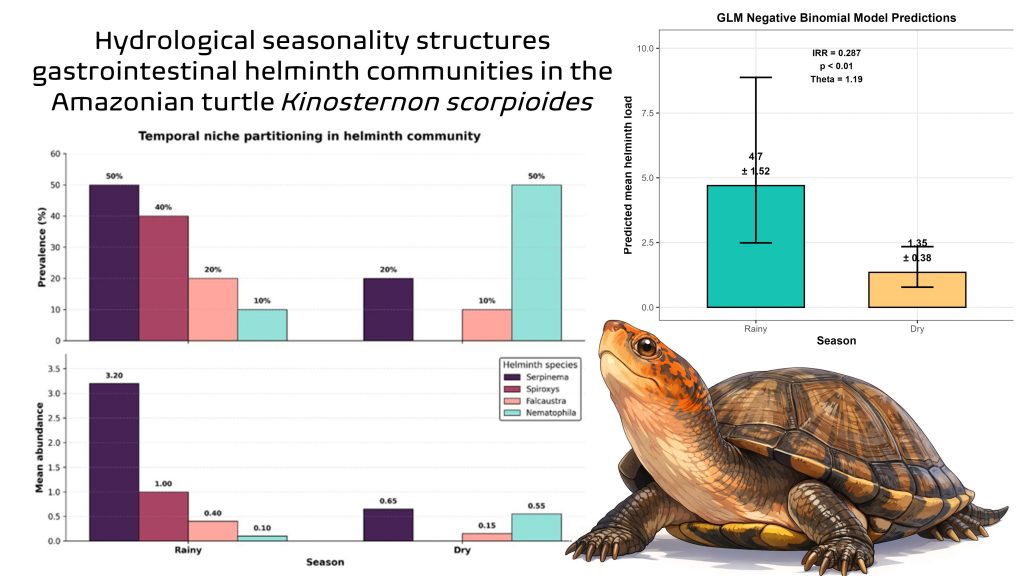
One of the most striking patterns involved Spiroxys figueiredoi. This species was found only during the rainy season and was completely absent during the dry season. By contrast, Nematophila grandis showed the opposite pattern, with higher prevalence in the dry season. Serpinema pelliculatus was the numerically dominant species, but without such an extreme seasonal shift.
We found this contrast fascinating because it suggested that species were not all responding to the same environmental filter. Some appeared to depend strongly on flooded conditions and on particular intermediate hosts; others seemed to benefit more from the concentration of organisms in shrinking water bodies during the dry season. This became a key point in the discussion, because it allowed us to move from a parasitological description to a more robust ecological interpretation.
In other words, we were not simply observing “parasites in turtles.” We were observing a community that reorganizes through time according to its transmission requirements.
Why we did not use a “regular” model
This was one of the most important methodological decisions in the study.
When we analyse parasite counts, the data rarely behave in a simple way. It is uncommon for all individuals to carry similar parasite loads. Instead, many hosts have few or no parasites, while a small number of individuals carry very high burdens. That pattern is known as aggregation or overdispersion, and it is very common in parasite ecology. The paper itself reports moderate aggregation, as indicated by the dispersion parameter of the negative binomial model.
That is why we did not use a “regular” Poisson regression. A Poisson model assumes a very strict relationship between the mean and the variance, and in this kind of dataset, that assumption often fails. If we had forced the data into that framework, we would have risked misrepresenting the system’s true variability.
Instead, we used a negative binomial GLM to test for seasonal differences in total helminth load. That choice allowed us to better model the aggregated structure of parasite counts and estimate the seasonal effect in a more biologically realistic way. The paper showed that individuals sampled during the dry season had a much lower expected parasite load than those sampled during the rainy season.
For us, this was not a purely technical choice. It was an ecological one. Choosing the right model meant respecting the system’s biology.
Looking beyond prevalence: seeing the whole community
Another thing we wanted to avoid was stopping at the classic parasitological metrics. Prevalence, mean abundance, and mean intensity are fundamental in parasitology, and we used them as defined by Bush et al. (1997). But we also knew that to understand how seasonality affects a parasite community, we needed to look further.
That is why we also examined richness, diversity, and community composition. And here we found something especially interesting: richness and diversity did not differ significantly between seasons, but community composition did differ significantly in the PERMANOVA. In other words, the number of species did not change much, but which species were present, and in what relative abundances, did change. The community did not simply become poorer or richer. It was reordered.
That nuance felt essential to us. In community ecology, a simple index is often insufficient to capture the underlying process. Two communities may have similar richness and still be ecologically very different.
What this study can offer future work
One of the things we find most exciting about this paper is that it opens both a methodological and a conceptual door.
Methodologically, it reinforces the idea that wildlife parasite studies gain a great deal when analysed with community ecology tools and models that match the actual distribution of the data. It is not only about identifying species or reporting prevalence. It is about understanding how species assemble, coexist, and respond to environmental change.
Conceptually, the study suggests that parasite communities can act as fine-scale ecological sensors. In this case, the helminth community responded to hydrological dynamics in a sensitive, structured manner. That means future studies could potentially use this kind of information to monitor altered flood pulses, changes in aquatic connectivity, or even early effects of climate change on Amazonian wetlands.
It also leaves us with valuable open questions.
Do these patterns repeat across years?
Do they occur similarly in other Amazonian turtle species?
How much do diet, body condition, or host immunity shape these seasonal differences?
The main conservation message
For us, this paper also carries a message that goes beyond parasitology.
The Amazon Basin is undergoing major transformations in its hydrological and climatic systems. The paper argues that changes in flood duration, rainfall patterns, or wetland connectivity may restructure parasite communities, favouring some species while disadvantaging others. And that matters because parasites are also part of biodiversity and ecosystem functioning.
One of the most powerful ideas in the paper is that Kinosternon scorpioides and its helminths may function as sentinel organisms. Monitoring parasite community composition could provide early signals of hydrological disruption or habitat degradation, complementing more traditional biodiversity monitoring approaches.
That is perhaps the conservation heart of the study: not seeing parasites only as a problem, but also as part of the ecological fabric that helps us read the condition of an ecosystem.
And yes, there is something controversial here
Bringing conservation and parasites into the same conversation is still uncomfortable for many people.
Parasites are often framed only in terms of disease, risk, or something that should be eliminated. But from an ecological perspective, that view is far too narrow. Parasites are part of biodiversity; they participate in food webs, respond to environmental change, and can act as sensitive indicators of ecological transformation. The paper explicitly takes up that point: parasite community composition may, in some cases, reflect environmental change more effectively than host abundance alone.
That does not mean romanticizing parasitism. It means accepting that if we want to understand complex ecosystems, we cannot leave out the uncomfortable organisms.
Behind the paper
On a personal level, this study reminded me of something that can easily get lost amid tables, scripts, and revisions: behind every analysis lies a way of seeing nature.
My role in the analysis and discussion pushed us in exactly that direction: to read the pattern carefully, to choose methods that matched the biology of the data, and to build an ecological narrative that did justice to the system’s complexity.
And perhaps that is the best summary of this paper: rainfall not only changes the landscape. It also reorganizes, in very precise ways, the invisible interactions that sustain a community.
Would you like to know more about this topic, or do you have something to say? I would be glad to hear from you. Feel free to get in touch.
Conga, D. F., Costa, J. H., Bejarano Alegre, V., Magalhães Bezerra, A., & Maciel de Castro Cardoso Jaques, A. (2026). Seasonal rainfall drives temporal niche partitioning in the helminth community of scorpion mud turtle (Kinosternon scorpioides) from Marajó Island. Journal of Helminthology. https://doi.org/10.1017/S0022149X25101132
————————————————————————————————————————————————————————————————–
Small capsules of the methods: why we used those analyses
1. Fisher’s exact test: We used this to compare prevalence between seasons, meaning whether the proportion of infected turtles changed between rainy and dry periods. It is well-suited for small sample sizes.
2. Negative binomial GLM: We used this to analyse total helminth load. We did not use a “regular” Poisson model because parasite counts were overdispersed: some individuals had many parasites, while others had very few. The negative binomial model handles that variation much better and captures parasite aggregation more appropriately.
3. Mann–Whitney U test: We used this to compare richness and diversity between seasons without assuming normality. It is a robust choice when data are not normally distributed or when sample sizes are limited.
4. PERMANOVA: We used this to test whether the composition of the helminth community changed between seasons. This was crucial because a community can contain a similar number of species and still differ strongly in which species are present and in their relative abundances.
5. Spearman correlation: We used this to test whether individuals with higher parasite richness also tended to have higher total parasite load. This helped us explore the relationship between co-infection and total burden.